 Healthline has strict sourcing guidelines and relies on peer-reviewed studies, academic research institutions, and medical associations. The vaccine doesn’t raise the risk of pregnancy complications. Studies have found that the whooping cough vaccine is safe for pregnant people and fetuses, according to the CDC. 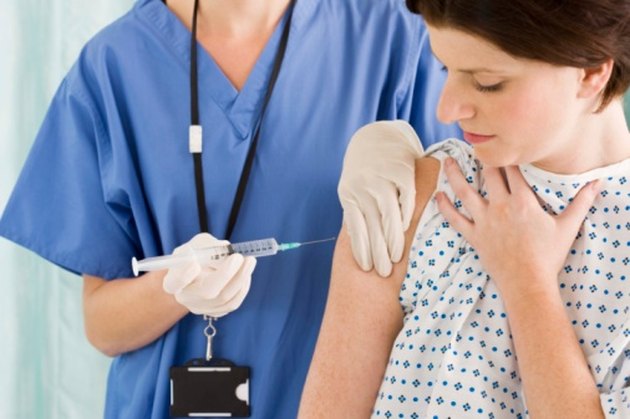
This will help protect the baby, after they’re born. If you’re pregnant, your body will pass these antibodies on to the fetus in your womb. The vaccine will cause your body to produce protective antibodies to help fight off whooping cough. To help protect young infants from whooping cough, the CDC advises pregnant adults to get the Tdap vaccine during the third trimester of pregnancy. Whooping cough can be very dangerous for young babies, and in some cases even fatal. That leaves them vulnerable to infection in the first months of life. If you’re pregnant, getting the whooping cough vaccine will help protect you and your unborn baby from the disease.Īlthough babies can be vaccinated against whooping cough, they typically get their first vaccine when they’re 2 months old. We found no evidence that DTP vaccine pertussis toxin plays a role in severe DTP vaccine reactions.Should you get the whooping cough vaccine in pregnancy? Vaccine endotoxin is a cause of febrile DTP vaccine reactions. 
Seizures associated with DTP vaccine have similar clinical characteristics as febrile seizures, and persistent crying is initiated by painful local reactions. No biologically active pertussis toxin was found in the acute sera of children experiencing severe DTP vaccine reactions. Some relatively elevated insulin values were noted however, this finding was also noted in the comparison group of children experiencing febrile seizures unrelated to immunization. Lymphocytosis did not occur, nor did hypoglycemia. Effects that may have been due to vaccine pertussis toxin were not found. Persistent crying was associated with painful local reactions. The children with seizures had a high rate of personal and family histories of seizures, and 90% had documented fevers (> or = 38 degrees C). Sixty children experienced severe reactions within 48 hours of DTP immunization: 32 children had seizures only, 14 subjects had hypotonic-hyporesponsive episodes, 2 subjects had fever > or = 40.5 degrees C only, 4 subjects had persistent crying > or = 3 hours, 6 children had seizures and fever > or = 40.5 degrees C, and 2 children had persistent crying and seizures. Serum was assayed for active pertussis toxin, both in free and immune-complex masked states. Blood was collected for complete blood cell count with leukocyte differential count, serum chemistry measurements, and insulin and glucose values. Follow-up examinations were performed 1 month later. Some comparisons were made between children with DTP vaccine-associated seizures and a comparison group of children experiencing febrile seizures unrelated to immunization.Ī history and physical examination were performed. Severe reactions included encephalopathy, persistent crying > or = 3 hours, hypotonic-hyporesponsive episodes (collapse episodes), fever > or = 40.5 degrees C, or seizures. Prospective, referral-based surveillance.Ĭhildren experiencing severe reactions within 48 hours of DTP immunization and evaluated within 24 hours of the reaction. To learn more about the causes of and risk factors for severe DTP vaccine reactions, children experiencing severe DTP vaccine reactions were studied. Large doses of pertussis toxin cause hyperinsulinemia and hypoglycemia as well as leukocytosis with a predominant lymphocytosis in animal models.  
Active pertussis toxin in DTP vaccine has been proposed to cause severe DTP vaccine reactions. The pathophysiology of severe reactions to diphtheria-tetanus-pertussis (DTP)vaccine is not well understood. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
